814
Views & Citations10
Likes & Shares
Medulloblastoma is one of the leading cancers in
pediatric patients. It is an embryonal tumor that originates from the
cerebellum or posterior fossa. It is one of the most common cancers in
children, and 40% cases were diagnosed below the age of 5 years. Data analysis
performed on surveillance, epidemiology and end-results (SEER) for 9 registries
suggested that 1372 cases were diagnosed in the period 1973 to 2007. The
associated risk factors for the development of medulloblastoma are unknown.
Pathophysiology can be best understood with the help of cellular signaling
pathways which includes signaling pathways like SHH Signaling, Wnt Signaling
and Notch Signaling. Till date, no immune-oncology product has been approved by
FDA for this indication. Some available immune-oncology products are under
investigation in clinical trials phase I to III. The complete perspective of
immunotherapy treatment has not been realized and utilized.
Keywords:
Medulloblastoma, Sonic Hedgehog (SHH) signaling, Wnt signaling, ErbB signaling,
C-myc signaling, IGF/PI3K signaling and nd notch signalling, Desmoplastic/nodular
type, Medulloblastoma with extensive nodularity, Large-cell variant, Anaplastic
medulloblastoma a, Environmental factors, Irradiation and radiation, Viral
infection, Genetic alteration, Monoclonal antibodies (mAbs), SHH inhibitors,
Tyrosine kinase autologous stem cell transplantation and autologous stem-cell
rescue
Abbreviations: AHSCR:
Autologous Stem-Cell Rescue; APC: Adenomatous Polyposis Coli; CBTRUS: Central
Brain Tumor Registry of the United States, CSF: Cerebrospinal Fluid; EGFR:
Epidermal Growth Factor; FDA: Food and Drug Administration; GSK3-β: Glycogen
Synthase Kinase 3-β; HDCT-AHSCR: High-Dose Chemotherapy with Autologous Stem
Cell Rescue; Hh: Hedgehog; IRS-1 Insulin Receptor Substrate-1; LRP: Lipoprotein
Receptor-Related Protein; LEF: Lymphoid Enhancer Factor; mAbs: Monoclonal
Antibodies; MAPK: Mitogen-Activated Protein Kinase; OSR: Overall Survival Rate;
PDGFRB: Platelet-Derived Growth Factor Receptor B, PI3K: Phosphatidylinositol
3-Kinase; PKB: Protein Kinase B; PTCH1: Gene Patched 1; mTOR: Mammalian Target
of Rapamycin, SEER: Surveillance, Epidemiology and End-Results; SHH: Sonic
Hedgehog Signaling; TCF: Transcription Factors T-Cell Factor, US: United States
INTRODUCTION
Medulloblastoma is
an embryonal tumor that originates from the cerebellum or posterior fossa [1].
Cerebrospinal fluid (CSF) plays an important role, in terms of metastasizing to
different sites in the brain and spine. Infratentorial tumors are invasive and
grow rapidly in comparison to other tumors [2].
Medulloblastoma is
one of the most common cancers in children and 40% cases were diagnosed below
the age of 5 years [3]. It accounts for less than 2% of adult brain tumors and
approximately 18% of pediatric brain tumors. The median age for medulloblastoma
is approximately 7 years in children and 25 years for adults [4].
Data analysis
performed on surveillance, epidemiology and end-results (SEER) for 9 registries
suggested that 1372 cases were diagnosed in the period 1973 to 2007. As per the
CBTRUS statistical report in 2005-2009, 2,617 cases of embryonal tumors was
identified [5].
Statistical representation of data in
accordance to gender, race and Hispanic ethnicity
A study conducted by
Padovani et al. [6] and similar studies in adults, stated that 5 years and 10
years overall survival rate (OSR) was 72% and 55% respectively, in 2004.
Etiology/predisposing factors
The associated risk factors for the
development of medulloblastoma are unknown. Familial history can increase the
chances of development of brain tumor.
Environmental factors: Diet and vitamins have
shown some effects due to the key role of nitrosamines, oxidants and
antioxidants. It is not clearly defined, but it might predispose to pediatric
brain tumor [8]. Some studies show effects of exposure to carcinogens as a
leading cause of pediatric brain tumor. Evidence revealed that the father
getting exposed to carcinogens prior to conception and the mother at the time
of maternity will damage DNA and predispose to cancer [9].
Irradiation and radiation: It has been
observed that children who are constantly exposed to radiations have higher
chances of developing childhood brain tumors. In a study, it was reported that
the lesser exposure to strontium-90 after the nuclear power plants ceased to
operate, led to lower incidences of brain tumor in children [10,11].
Viral infection: Few studies suggested a
strong relationship between human neurotropic polyoma virus, i.e., JC virus and
medulloblastoma. Viral protein plays an important role in the binding efficacy,
interference to tumor suppressor activity and regulation of protein like p53
and Rb [12]. Exposure to measles immunization and SV40 shows link to the
development of medulloblastoma [13].
Genetic alteration: Various genetic disorders
act as important predisposing factor for the development of medulloblastoma.
These syndromes along with their genes and chromosomal locations have been
mentioned in Table 2.
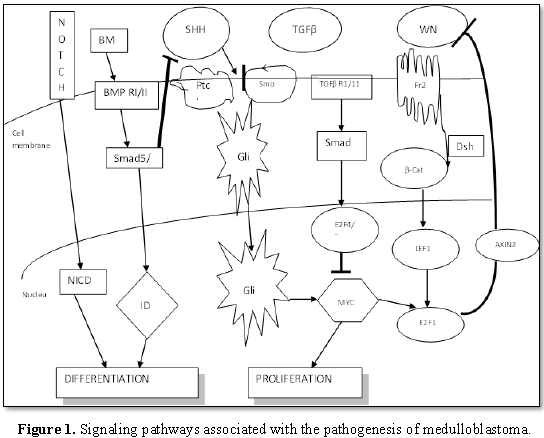
Pathophysiology
and molecular basis
Pathophysiology
can be best understood with the help of cellular signaling pathways.
Figure 1 explains the
various signaling pathways associated with the pathogenesis of medulloblastoma.
Sonic Hedgehog (SHH) signaling
Gene Patched 1
(PTCH1), a tumor suppressor, located on 9q22.3 chromosomal location encrypts
for Hedgehog proteins’ transmembrane surface receptor. This pathway is
associated with the progression of external granular layer of the cerebellum.
Purkinje cells produce SHH, which binds to PTCH1 receptor and makes the
smoothened (SMO) free from inhibition by activating the proliferation of
precursors of cerebellar granule cell. It also activates the Gli family of
transcription factors. In medulloblastoma, BMI1 is over-expressed. This
disrupts the normal regulation of signaling pathways like Rb and p53 [14].
Wnt signaling
A protein
complex is formed by adenomatous polyposis coli (APC) in association with
β-catenin (CTNNB1), axin 1 (AXIN1) and glycogen synthase kinase 3-β (GSK3-β).
In case of Turcot’s syndrome, APC is germ line mutated and is the predisposing
factor for the development of medulloblastomas [15].
Wingless (WNT)
ligand binds to a receptor complex, which includes seven transmembrane Frizzled
(FZ), serpentine receptor and low density lipoprotein receptor-related protein
(LRP). CTNNB1 is prevented from phosphorylation by glycogen synthase kinase-3β
(GSK-3β) and is translocated to the nucleus. Upon activation, a downstream
effect of β-catenin is observed and deprivation is followed by TCF
(Transcription Factors T-cell Factor)/LEF (Lymphoid Enhancer Factor)
interaction with activation of transcription of Wnt targets gene (c-Myc, cyclin
D1 and AXIN2) [16].
Survivin, an
apoptosis inhibitor is also over-expressed in the presence of activation of Wnt
signaling pathway. SOX4 and SOX11 are over-expressed as well and are linked to
medulloblastoma [15].
Notch signaling
In Human, 4
types of NOTCH receptor have been identified. Notch, heterodimeric receptor, is
a single transmembrane protein. NOTCH 2 is overexpressed in case of
medulloblastoma. Extracellular domain and cytoplasimc domain contains different
binding efficacy. Cytoplasmic domain contains domain like RAM, a transcription
transactivation, two nuclear localization signals, six CDC10 repeats and a PEST
sequence. Extracellular binding helps in ligand binding and contains epidermal
growth factor (EGFR) like repeats. In the absence of ligand binding,
extracellular domain will impede signaling. Once NOTCH binds to ligands Jagged
(JAG-1, JAG-2) and Delta-like (DLL-1, DLL-2, DLL-3) family members, release of
Notch intracellular domain (NCI) and translocation to the nucleus takes
place.[16] NCI interacts with DNA binding proteins (CBF1), which in turn
activate the loop-helix transcription factors HES1 and HES5. HES1 forms
transcriptional repressor complexes with FOXG1 which negatively regulates the
differentiation of neural progenitor cells [15].
ErbB signaling
ErbB include
four receptors (ErbB1-ErbB4) and a variety of ligands and neroregulins. They
belong to tyrosine kinase family. ErbB4, CYT1 isoform, is over-expressed in
tumor, which activates anti-apoptotic phosphatidylinositol 3-kinase
(PI3K)/protein kinase B (PKB)/AKT signaling. ErbB2 gene is located on
17q11.2-q12 chromosome and is regarded as a prospective medulloblastoma
oncogene. The ErbB ligand, NRG1-β is expressed by ErbB2 and ErbB4 results in
disease progression, while C-myc is the leading target.
C-myc signaling
C-myc is linked
to the activation of the different signaling pathways like SHH and Wnt
pathways, translocations, viral insertion, genomic amplification and activating
mutations. C-myc binds to JPO2 protein, which can activate C-myc
transformation. It is related to metastatic medulloblastoma. N-Myc is linked to
SHH signaling pathway. PI3K prevents the degradation of N-Myc and in turn,
enhances the effects of IGF/PI3K signaling pathway. This process explains the
development of medulloblastoma associated with SHH pathway.
IGF/PI3K signaling
IGF-1 receptor
(IGF-1R) protein (e.g. insulin receptor substrate-1 (IRS-1), PI3K, AKT/PKB,
Erk-1 and Erk-2) and activated phosphorylated form of IGF-1R are
over-expressed, in case of medulloblastoma. Inhibition of dephosphorylation of
GSK3-β and IGF-1R are important for the management of medulloblastoma by
reducing tumor growth. IGF-1R signaling pathway can lead to the activation of
AKT, PI3K, ras/MAPK (mitogen-activated protein kinase) signaling. In case of
metastatic medulloblastoma, up-regulation of ras/MAPK pathway and
platelet-derived growth factor receptor B (PDGFRB) are key factor [17].
CELLS OF ORIGIN
Activation of
different signaling pathways in different medulloblastoma subtypes suggest that
medulloblastomas have different origins. There are four types of
medulloblastoma identified, based on the following molecular and genetic
aspects as well as clinical and prognostic features.
1) Desmoplastic/nodular
type
2) Medulloblastoma
with extensive nodularity
3) Large-cell
variant
4) Anaplastic
medulloblastoma
IMMUNOTHERAPY
Monoclonal antibodies (mAbs)
Non-FDA
approved monoclonal antibodies: There is no FDA approved monoclonal antibody as
an immunotherapy for medulloblastoma. However, Bevacizumab is under clinical
trials for the management of medulloblastoma as mentioned below in Table 3.
SHH inhibitors:
Non-FDA approved SHH inhibitor: Currently no SHH inhibitor is approved by FDA for medulloblastoma.
However, some SHH inhibitors that are under clinical trials are mentioned in Table 4. The secreted SHH ligand
generate the signal by smoothened (SMO) receptor and helps in the proliferation
of neural precursor cells.
Tyrosine
kinase inhibitors
Non-FDA approved tyrosine kinase inhibitors: Currently, no tyrosine kinase inhibitor is approved by FDA for
medulloblastoma. However, the kinase inhibitors under clinical trials are
mentioned in Table 5.
Autologous stem cell transplantation
Autologous stem-cell rescue: The procedure of high-dose chemotherapy with autologous stem cell
rescue (HDCT-AHSCR) has been one of the successful treatment modalities in
medulloblastoma. AHSCR also restores the suppression of the process of
hematopoiesis, which is limited by the dose of the chemotherapy [24].
A COG trial has
been done using the combination of chemotherapy and peripheral stem cell
transplant. This COG trial is open for children aged 3 years or younger, on
being diagnosed with high-risk disease, which is defined as those with
disseminated and/or sub-totally resected tumors or those younger than 8 months
with otherwise standard-risk disease. Patients with cortical primitive
neuroectodermal tumors or pineoblastomas are also eligible. This study is
evaluating chemotherapy as given in the completed COG study COG-99703, which
used multi-agent chemotherapy followed by thiotepa-based, higher-dose,
marrow-ablative chemotherapy and peripheral stem cell rescue, and randomly
assigns patients to treatment with or without intravenous high-dose
methotrexate [25].
COX-2 inhibitors
Non-FDA
approved COX-2 inhibitors: Currently, no COX-2 inhibitor is approved by FDA for
medulloblastoma. However, the COX-2 inhibitors under clinical trials are mentioned
in Table 6.
mTOR
inhibitors
Non-FDA approved mTOR inhibitors: Currently, no mTOR inhibitor is approved by FDA for medulloblastoma.
However, the mTOR inhibitors under clinical trials are mentioned in Table 7.
CONCLUSION
Medulloblastoma
is one of the leading cancers in pediatric patients. Pathophysiology includes
signaling pathways like SHH Signaling, Wnt Signaling and Notch Signaling. Apart
from that, chromosome 17, p53, 17p gene, RENKCTD11, MnT and duplication of
genes on 17q are few leading causes for the pathogenesis of medulloblastoma.
Till date, no immune-oncology product has been approved by FDA for this
indication. Some available immune-oncology products are under investigation in
clinical trials phase I to III. The complete perspective of immunotherapy
treatment has not been realized and utilized. Proper pre-clinical and clinical
designs are the important pillars in understanding the future of immunotherapy
in treating cancer patients.
1. http://www.williamdoolin.com/what-is-medulloblastoma.html
2. Okamoto
Y, Shimizu K, Tamura K, Miyao Y, Yamada M, et al. (1988) An adoptive
immunotherapy of patients with medulloblastoma by lymphokine-activated killer cells
(LAK), ActaNeurochir (Wien) 94: 47-52.
3. http://wikitheme.warmachine.im/Medulloblastoma
4. Smoll
NR, Drummond KJ (2012) The Incidence of medulloblastomas and primitive
neuroectodermal tumors in adults and children. J Clin Neurosci 19: 1541-1544.
5. CBTRUS
(2005-2009) Number of brain and central nervous system tumors by major
histology groupings, histology, gender, race and Hispanic ethnicity. CBTRUS
Statistical Report: NPCR and SEER.
6. Padovani
L, Sunyach MP, Perol D, Mercier C, Alapetite C, et al. (2007) Common strategy
for adult and pediatric medulloblastoma: A multicenter series of 253 adults.
Int J Radiat Oncol Biol Phys 68: 433-440.
7. Farwell
JR, Dohrmann GJ, Flannery JT (1984) Medulloblastoma in childhood: An
epidemiological study. J Neurosurg 61: 657-664.
8. Wrensch
M, Minn Y, Chew T, Bondy M, Berger MS (2002) Epidemiology of primary brain
tumors: Current concepts and review of the literature. Neuro Oncol 4: 278-299.
9. Wrensch
M, Lee M, Miike R, Newman B, Barger G, et al. (1997) Familial and personal
medical history of cancer and nervous system conditions among adults with
glioma and controls. Am J Epidemiol 145: 581-593.
10. Solomon
G; Natural Resources Defense Council (2014). Brain and central nervous system
tumors: The collaborative on health and the environment. Available from: http://www.healthandenvironment.org/brain_cancer/peer_reviewed
11. Mangano
JJ, Gould JM, Sternglass EJ, Sherman JD, Brown J, et al. (2002) Infant death
and childhood cancer reductions after nuclear plant closings in the United
States. Arch Environ Health 57: 23-31.
12. Caracciolo
V, Reiss K, Khalili K, De Falco G, Giordano A (2006) Role of the interaction
between large T antigen and Rb family members in the oncogenicity of JC virus.
Oncogene 25: 5294-5301.
13. Farwell
JR, Dohrmann GJ, Flannery JT (1984) Medulloblastoma in childhood: An
epidemiological study. J Neurosurg 61: 657-664.
14. de
Bont JM, Packer RJ, Michiels EM, den Boer ML, Pieters R (2008) Biological
background of pediatric medulloblastoma and ependymoma: A review from a
translational research perspective. Neuro Oncol 10: 1040–1060.
15. Li
KK, Lau KM, Ng HK (2013) Signaling pathway and molecular subgroups of medulloblastoma.
Int J Clin Exp Pathol 6: 1211-1222.
16. Kim
J, Rudin CM (2014) Hedgehog Signaling Pathway: Therapeutic abrogation of a
fundamental developmental molecular pathway, molecular marker. ASCO Meetings.
Available from: http://am.asco.org/hedgehog-signaling-pathway-therapeutic-abrogation-fundamental-developmental-molecular-pathway
17. National
Cancer Institute (NCI) (2014) Emozolomide and irinotecan hydrochloride with or
without bevacizumab in treating young patients with recurrent or refractory
medulloblastoma or CNS primitive neuroectodermal tumors. In: ClinicalTrials.gov
[Internet].
18. Novartis
Pharmaceuticals (2014) A Phase II Study of Oral LDE225 in patients with
hedgehog (Hh)-pathway activated relapsed medulloblastoma (MB). In:
ClinicalTrials.gov [Internet].
19. Sholler
G (2014) Spectrum Health Hospitals. Erivedge (Vismodegib) in the treatment of
pediatric patients with refractory pontine glioma. In: ClinicalTrials.gov
[Internet
20. Lilly
E (2014) A study of LY2940680 in pediatric medulloblastoma or rhabdomyosarcoma.
In: ClinicalTrials.gov [Internet].
21. National
Cancer Institute (NCI) (2014) Lapatinib in treating young patients with recurrent
or refractory central nervous system tumors. In: ClinicalTrials.gov [Internet].
22. National
Cancer Institute (NCI) (2014) Erlotinib and temozolomide in treating young
patients with recurrent or refractory solid tumors. In: ClinicalTrials.gov
[Internet].
23. Panosyan
EH, Ikeda AK, Chang VY, Laks DR, Reeb CL, et al. (2011) High dose chemotherapy
with autologous hematopoietic stem-cell rescue for pediatric brain tumor
patients: A single institution experience from UCLA. J Transplant 2011: 1-11.
24. Seoul
National University Hospital, Seoul National University Hospital (2014) Tandem
high dose chemotherapy and autologous stem cell rescue for high risk pediatric
brain tumors. In: ClinicalTrials.gov [Internet].
25. Helsinki
University Central Hospital (2014) ANGIOCOMB anti-angiogenic therapy for
pediatric patients with diffuse brain stem and thalamic tumors. In:
ClinicalTrials.gov [Internet].
26. Emory
University; Children's Healthcare of Atlanta (2014) Aflac ST0901 CHOANOME -
Sirolimus in solid tumors. In: ClinicalTrials.gov [Internet].